
Different risks must be taken into consideration. It is up to the pharmaceutical user to make a risk-based decision. For sure is that no GMP guideline requires the one or the other certificate. In the end, the answer is always "it depends". 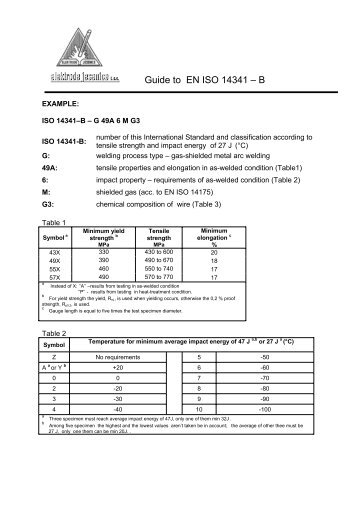
The frequent question arising is whether a 2.2 or a 3.1 certificate is needed, as 3.1 certificates are more expensive. A link to each component with 3.1 certificates to the respective batch/melt is available. A person independent from the manufacture is in charge of the test report. The performance of the tests is fixed in technical rules or in the specification/order. Test certificate 3.1 - The tests are determined in advance.Certificate 2.2 - Same as 2.1 with additional indication of the test results based on non-specific inspection and testing (the manufacturer determines the tests himself).Certificate 2.1 - confirmation from the manufacturer that the product supplied complies with the requirements of the order.Still, this topic is continuously being discussed and can be found in respective specialised forums.Īccording to DIN-EN 10204, the following applies: 
The question around the material certificates needed for metallic products to comply with GMP documentation has long existed. 
GMP Courses & Conferences on Site (in hotels).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
